
 By Jon Nelson, BHI Director of Client Engagement: The SBIR/STTR program is currently paused pending reauthorization. However, there is a light at the end of the tunnel. On April 2nd, the Small Business Innovation and Economic Security Act was presented to the President for his signature. Three different outcomes now lay before us.
By Jon Nelson, BHI Director of Client Engagement: The SBIR/STTR program is currently paused pending reauthorization. However, there is a light at the end of the tunnel. On April 2nd, the Small Business Innovation and Economic Security Act was presented to the President for his signature. Three different outcomes now lay before us.
- The President could sign the bill into law.
- The President may choose to neither sign, nor veto the bill, in which case, the bill will automatically become law on April 14th.
- The President could veto the bill. In this situation, the bill would return to Congress, where a two-thirds majority would be needed in both houses in order to override the veto.
For companies that have been relying on federal funding as part of their near-term financing strategy, the pause is a genuinely difficult disruption. Plans are delayed, timelines shift, and the natural response is to wait until there is more clarity especially when there are countless other priorities competing for attention. That instinct is understandable. However, the teams that emerge from this period in the strongest competitive position will be the ones that resist it.
While this is the first lapse in the SBIR/STTR program since its inception in the 1980s, other large funding opportunities and federal programs have seen similar pauses. Thus, history is instructive here – when a high-demand funding program goes dark and then reopens, submission volume typically reflects the backlog that accumulated during the gap. Companies that were mid-preparation when the pause began, teams that used the intervening time to get ready, and applicants who hesitated will all enter the queue at roughly the same time.
The result is a more competitive review environment. The bar for what constitutes a competitive proposal is effectively higher than it would be in a typical cycle, and where the difference between a thoughtfully developed application and one that was assembled quickly becomes far more apparent for reviewers to see.
What Good Preparation Actually Looks Like
For most applicants, the limiting factor in a competitive submission is not the quality of the underlying science. It is how clearly the proposal communicates that science to a review panel, how convincingly it is positioned within a credible commercialization strategy, and how clearly the proposal demonstrates that this team, with this approach, at this stage of development, is worth funding. Each of those elements takes longer to develop than most first-time applicants expect, and they each suffer when compressed into the final weeks before a deadline.
A strong commercialization narrative requires that holds up under reviewer scrutiny goes through multiple rounds of drafting and refinement. A technical approach that translates well on the page requires careful editing by individuals who understand both the science and reviewer expectations. A budget that avoids unnecessary scrutiny is one that has been reviewed with agency expectations and common pitfalls in mind.
The best time to do this work is now, before the pressure of an open submission window makes careful development difficult.
Getting the Most Out of the Time Available
Beyond the core proposal materials, there is meaningful preparatory work that is often deferred under deadline pressure. Reviewing and organizing preliminary data, identifying the most relevant solicitations to target when the program reopens, aligning internal stakeholders on project scope and budget, and stress-testing the overall narrative against likely reviewer questions are all tasks that benefit from careful attention that is hard to give them when a submission deadline is just around the corner.
This is also the right time to pursue the relationship-driven components of a strong application. Securing letters of support from key stakeholders, whether from clinical partners, academic collaborators, or potential customers, takes time and follow-up, and letters that are clearly written with care carry more weight with reviewers than ones that read as last-minute requests.
Similarly, identifying and formalizing relationships with contract research organizations or other external partners strengthens both the technical credibility of the proposal and the team’s demonstrated capacity to execute. These conversations take time , and teams that have already invested in them are at a clear advantage in a when the submission window reopens.
Working with BioHealth Innovation
At BioHealth Innovation, we work with early-stage founders and research teams throughout the full proposal development process, from identifying the right funding opportunity to building the commercialization narrative and finalizing technical and budget documents.
Teams that engage early enter submission cycles significantly more prepared. In a competitive environment, that head start can make big difference.
If you are serious about competing when the SBIR/STTR program reopens, the right time to start is now.
Contact us at jnelson@biohealthinnovation.org


 By Catherine Leasure, Ph.D., BHI Life Sciences Business Strategist – If you’ve written a grant recently, you’ve probably wondered whether AI could make the process easier. Maybe you’ve already tried it. The honest answer is that AI can help, but how much depends entirely on what you bring to it. When you know what you’re doing, it gets you to a solid draft faster. However, without a strong grasp of the process behind it, it can produce polished-sounding text that misses the mark in ways that aren’t always obvious until a reviewer or experienced grant writer points them out.
By Catherine Leasure, Ph.D., BHI Life Sciences Business Strategist – If you’ve written a grant recently, you’ve probably wondered whether AI could make the process easier. Maybe you’ve already tried it. The honest answer is that AI can help, but how much depends entirely on what you bring to it. When you know what you’re doing, it gets you to a solid draft faster. However, without a strong grasp of the process behind it, it can produce polished-sounding text that misses the mark in ways that aren’t always obvious until a reviewer or experienced grant writer points them out.

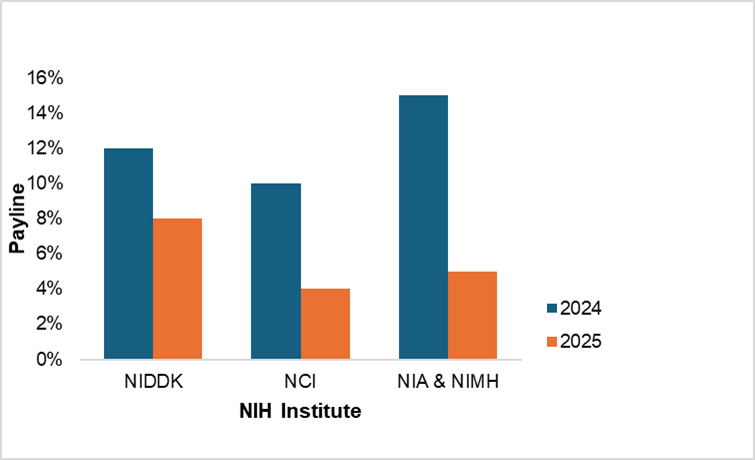 In practice, this means that instead of spreading the cost of a three-year grant across three annual appropriations, NIH must now allocate the full amount from the current year’s budget. As a result, far fewer grants can be awarded.
In practice, this means that instead of spreading the cost of a three-year grant across three annual appropriations, NIH must now allocate the full amount from the current year’s budget. As a result, far fewer grants can be awarded.