

University System of Maryland Chancellor William E. Kirwan, who announced in the spring his plans to step down, said this week that he will likely remain in his post at least through February as the search continues for his successor.


University System of Maryland Chancellor William E. Kirwan, who announced in the spring his plans to step down, said this week that he will likely remain in his post at least through February as the search continues for his successor.
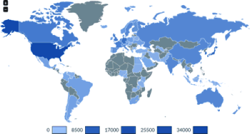

As an offshoot of PatentStat, I’ve assembled a set of graphs of patent inventor locations, focusing in 33 industry sectors.


Bedsores, diabetic ulcers and other chronic wounds cost the U.S. health care system $30 billion a year. Why? At least in part because the primary tool doctors and nurses use to track wounds is a basic ruler.
A ruler can measure the size of a wound, but does little to track other important qualities, such as changes in shape and tissue color. Consider that patients are usually cared for by a rotating team of nurses, who may each interpret a wound’s appearance differently, and it’s easier to see how so much money is spent tending to preventable (or at least treatable) conditions.
![]()
Recent efforts between the University of Maryland (UMD) and Bethesda-based Weinberg Medical Physics LLC (WMP) have led to a new technique to magnetically deliver drug-carrying particles to hard-to-reach targets. The method has the potential to transform the way deep-tissue tumors and other diseases are treated.


Pieris AG announced today the initiation of a Phase I clinical trial with PRS-080, an anti-hepcidin Anticalin® therapeutic protein designed to treat anemia. The trial is a placebo-controlled, single ascending dose evaluation of the compound’s safety and tolerability in healthy volunteers. Conducted in Germany, the trial is underway and patients from the first cohort have been dosed.


When it comes to the biggest investors in Research and Development (R&D) Swiss pharma top dogs Novartis and Roche have defended their positioning among the top ten companies worldwide – according to a recently published EU study.
The University of Maryland is now integrating the L![]() ean LaunchPad® into standard innovation and entrepreneurship courses across all 12 colleges within the University. Over 44 classes have embedded the business model canvas and/or Customer Discovery including a year-long course taken by every single one of its bioengineering majors.
ean LaunchPad® into standard innovation and entrepreneurship courses across all 12 colleges within the University. Over 44 classes have embedded the business model canvas and/or Customer Discovery including a year-long course taken by every single one of its bioengineering majors.


Aris Melissaratos is batting around the idea of making Stevenson University’s Maryland Rising economic development forum an annual event.
Can you blame him? The first conference, held Monday, drew big-name speakers and generated a buzz other economic development confabs probably envy.


Hospitals are on the hunt for new ideas to transform health care, and some are looking to reality television for inspiration.
Several health care institutions recently have tested TV’s “Shark Tank” approach to seeking out innovation — gathering a panel of pros to hear invention ideas from startups.


Funding and Research Opportunities
The following funding opportunity announcements from the NHLBI or other components of the National Institutes of Health, might be of interest:
Notices:
Requests for Applications: