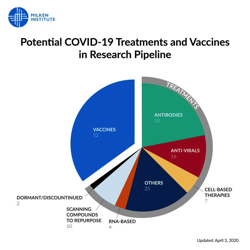

We’re currently tracking 95 potential treatments and 52 potential vaccines in the pipeline to combat hashtag#COVID19.
There are thousands of researchers all over the world racing to get a treatment or vaccine to market.


We’re currently tracking 95 potential treatments and 52 potential vaccines in the pipeline to combat hashtag#COVID19.
There are thousands of researchers all over the world racing to get a treatment or vaccine to market.


The NIH is deeply concerned for the health and safety of people involved in NIH research, and about the effects on the biomedical enterprise in the areas affected by the HHS declared public health emergency for COVID-19. Due to the potential exceptional impact, we want to assure our recipient community that NIH will be doing our part to help you continue your research.


SAN FRANCISCO–(BUSINESS WIRE)– venBio Partners LLC today announced the closing of venBio Global Strategic Fund III (“venBio Fund III”), its third life sciences venture capital fund, exceeding its target and closing on approximately $394 million in capital commitments in an oversubscribed fundraise. The capital was raised from existing and new investors, including a broad range of institutional investors comprising pharmaceutical companies, corporate pensions, financial institutions, endowments and foundations, family offices and funds-of-funds.


ROCKVILLE, Md., April 6, 2020 /PRNewswire/ — Emmes today announced that Heather Hill, Dr. Adam Mendizabal and Dr. Nilay Shah have been promoted to vice president. According to Dr. Anne Lindblad, president and chief executive officer, “These promotions reflect their talent and value to the organization. Heather, Adam and Nilay demonstrate our commitment to developing staff and promoting from within. They will strengthen and add great value to our leadership team and will support our plans to grow both internally and externally.”
Heather Hill
Dr. Adam Mendizabal
Dr. Nilay Shah


One of America’s biggest companies has teamed up with one of the country’s least-known federal agencies to make doses of a coronavirus vaccine. Lots of them.
The deal underscores the important role that the U.S. Biomedical Advanced Research and Development Authority, or BARDA, plays behind the scenes in protecting Americans—and everyone, really—from pandemics.

A few weeks ago, I was in New Orleans for a jam-packed wedding weekend, complete with a crawfish boil, second line parade, Hawaii-themed 30th birthday luau for the bride-to-be, and more gumbo and jambalaya than we could eat (and guys, we’re talking authentic Louisiana Creole, which was just delicious.)
few weeks ago, I was in New Orleans for a jam-packed wedding weekend, complete with a crawfish boil, second line parade, Hawaii-themed 30th birthday luau for the bride-to-be, and more gumbo and jambalaya than we could eat (and guys, we’re talking authentic Louisiana Creole, which was just delicious.)
Image: Image via the Business Journals

Last week, we spoke to the head of Emergent BioSolutions’ Therapeutics Business Unit Dr. Laura Saward about her company’s work developing plasma-based potential treatments for COVID-19. Now, the company announced that it has received $14.5 million in funding from the Biomedical Advanced Research and Development Authority (BARDA), which is part of the U.S. Department of Health and Human Services (HHS), to speed the development of one of its treatment candidates.


The U.S. Food and Drug Administration approved a clinical trial Friday that will allow Johns Hopkins University researchers to test a therapy for COVID-19 that uses plasma from recovering patients.
Arturo Casadevall, a Johns Hopkins infectious disease expert, proposed the use of convalescent plasma to treat critically ill COVID-19 patients and to boost the immune systems of health care providers and first responders. He assembled a team of physicians and scientists from around the United States to establish a network of hospitals and blood banks that can collect, isolate, and process blood plasma from COVID-19 survivors


COVID-19 Notice for Maryland SBDC Clients, Stakeholders, and Partners
We are committed to serving our clients during the COVID-19 (Coronavirus) health emergency.
Consulting Services – New clients already in business can request COVID consulting assistance by clicking on the “I’M IN BUSINESS- GET CONSULTING” on our home page. The link is right below this notice towards the right side of the page. Continuing clients may continue to contact their consultant by phone, web conference, or email.
Training – All in-person training scheduled during the month of April has been canceled, postponed or offered online. If a paid training class is canceled or postponed, participants may request a refund or apply the funds to another class. If the class will be offered online, registered participants will receive an email with instructions for participating in the online class.


We’re proud to announce a partnership with the U.S. Government on a comprehensive response to expedite development of our plasma-derived therapy to address hashtag#COVID19. https://lnkd.in/eHGC5F6
hashtag#toprotectandenhance hashtag#emergentbiosolutions hashtag#COVID19 hashtag#coronavirus
Image: https://www.linkedin.com