

Led by Noble Capital Markets and Tichenor Ventures to Fund OST-HER2 PhIIb Trial and OST-TDC Preclinical Next Generation ADC
CAMBRIDGE, Md., April 20, 2021 /PRNewswire/ — OS Therapies, a clinical-stage biopharmaceutical company focused on discovering and developing innovative therapies to treat and cure Osteosarcoma (OS) and other deadly cancers in kids and adults including ovarian, esophageal, endometrial and lung cancers, today announced the closing of a $6 million Series A round of funding.


The purpose of this event is to bring Technology Transfer leaders, policy makers, thought leaders, and innovators in the Technology Transfer field together to discuss and exchange ideas on how to best qualify and quantify the impact of Technology Transfer on the stakeholders it serves.


ROCKVILLE, Md., April 20, 2021 /PRNewswire/ — Cellphire Therapeutics, Inc., a biotechnology company developing next-generation platelet-based hemostatic therapeutics for application across multiple medical indications, announced today that Michael Gaffney has joined the company as Chief Executive Officer, effective immediately. Mr. Gaffney brings deep health sector insight, extensive operational experience, and proven strategy and capital markets expertise to Cellphire. He will also join the Company’s board.


The Montgomery County Economic Development Corporation (MCEDC) and the Federal Laboratory Consortium (FLC) have signed a Memorandum of Understanding (MOU) to formalize a strategic partnership to help build a stronger local relationship between federal labs and Montgomery County’s private sector, particularly small businesses.
![]()
COLUMBIA, Md. (April 20, 2021)—TEDCO, Maryland’s economic engine for technology companies, announced today its Seed Fund invested $200K in TargetDocs, a Maryland-based technology startup. TEDCO’s Seed investment is part of an $850K funding round, which includes University System of Maryland Momentum Fund (Momentum Fund), the Hustle Fund, New Age Capital, Debut Capital, Dingman Center Angels, and Gaingels.
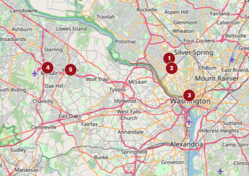

Largest Venture Capital Investors in Greater D.C. – Washington Business Journal
Image: https://www.bizjournals.com


U.S.-listed shares of GlaxoSmithKline GSK, 0.12% were down 1.4% in trading on Thursday after the Food and Drug Administration said it had granted accelerated approval to the company’s endometrial cancer treatment. The immunotherapy, which was called dostarlimab and is now named Jemperli, is approved to treat patients with severe forms of this kind of cancer who have a genetic feature known as dMMR.


Amid the international effects of the COVID-19 pandemic, Maccura Biotechnology, headquartered in the city of Chengdu in China’s Sichuan Province, is still able to keep up with changing demands. The Maryland Department of Commerce first met with the well-established, global diagnostics company in 2018 with the help of the Maryland Center China.


BETHESDA, Md., April 20, 2021 (GLOBE NEWSWIRE) — Gain Therapeutics, Inc. (NASDAQ: GANX) (“Gain”) today announced a multi-target collaboration agreement with Zentalis Pharmaceuticals (NASDAQ: ZNTL) to discover new product candidates for the treatment of cancer. Gain will use its proprietary SEE-Tx computational platform technology to identify new sites on target proteins for potential use in oncology. SEE-Tx applies a proprietary computational algorithm and supercomputer processing to the published 3D structure of proteins to discover new binding sites with the ability to modulate protein function. The intended output is newly-discovered targets or target protein interactions that can then be drugged for therapeutic benefit to intervene on protein misfolding.
![]()
RESEARCH TRIANGLE PARK — United Therapeutics has applied for a priority review voucher and submitted a new drug application to the United States Food and Drug Administration, the company announced this week. The drug, Tyvaso DPI™, is a novel dry powder inhalation formulation of treprostinil, and, if approved, will be used to treat pulmonary arterial hypertension (PAH).
The company estimates there are about 30,000 treatable patients in the United States, according to a statement.
