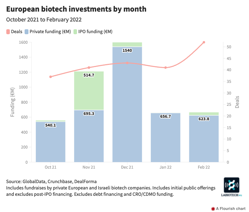

Companies developing lab automation, ophthalmological treatments, and novel foods received Europe’s top biotech investments in February 2022.
European and Israeli biotech investments in February 2022 narrowly beat January’s performance. Life sciences companies in these regions bagged 52 private investments and initial public offerings (IPOs), worth €664M in total. Meanwhile, January’s tally was 41 deals totaling €660M. Neither month matched the biotech investment highs of December 2021.
Image: https://www.labiotech.eu