|

Governor Martin O’Malley today announced that Rachel King, co-founder and CEO of GlycoMimetics in Gaithersburg, has been named chair of the Maryland Life Sciences Advisory Board (LSAB). King will replace chair H. Thomas Watkins, former President and Chief Executive Officer of Human Genome Sciences, Inc., who has served on the Board since Governor O’Malley and the Maryland General Assembly created it in 2007. As chair, Watkins led the Board through a strategic planning process that, working closely with Governor O’Malley, resulted in BioMaryland 2020, a 10-year, $1.3 billion strategy for moving Maryland’s life sciences industry forward.
back to top 

The Economic Alliance of Greater Baltimore (EAGB) and Maryland Department of Business of Economic Development (DBED) have announced the creation of Advance Maryland, a business program designed to support growth companies, an integral component in the prosperity and sustainability of local economies. Advance Maryland was established to provide resources targeted to second-stage companies. These companies are growth-oriented and have moved beyond the startup phase. They are at the forefront of job creation and critical to vibrant economies. In contrast to traditional business assistance which focuses on finances, business plans and operational issues,
Advance Maryland addresses strategic growth challenges, from developing new markets and refining business models, to gaining access to competitive intelligence. “Maryland has a plethora of organizations and resources devoted to the startup community, but we are limited when it comes to resources for second stage companies. The statistics show how essential it is to recognize these companies and make the necessary tools available to support their growth,” stated Jen Gunner, COO of EAGB and Co-Program Manager of Advance Maryland. Youreconomy.com states that between 1995 and 2009, second-stage companies represented eleven percent of U.S. establishments, but generated more than thirty six percent of jobs and thirty eight percent of sales.
back to top 

University of Maryland (UM) Ventures and SilcsBio, LLC announced today that SilcsBio has obtained exclusive rights to a technology licensed from the University of Maryland, Baltimore (UMB). UM Ventures is an ambitious joint research commercialization effort of the UMB and the University of Maryland, College Park (UMCP). SilcsBio is a supplier of computer-directed drug discovery software and services.
"The license, which we obtained from UMB, creates the core of our product line," said Kelli Booth, SilcsBio's Chief Operating Officer. "It's great to have a university so supportive of our state's start-up community."
back to top 

The Chinese Biopharmaceutical Association, USA (CBA) will host the 18th Annual Conference at the University of Maryland Shady Grove Conference Center on Saturday, June 15th, 2013. The theme of this year’s conference is “Global Partnership in Biopharmaceutics and Translational Medicine;” it will address the critical importance of establishing worldwide collaboration to capture the great opportunity for the advancement of modern medicine and biopharmaceuticals.
The Conference
The Conference includes five sessions:
- Drug Discovery: New Strategies and Platforms
- Translational Genomics and anti-cancer therapy
- Opportunities in New high tech parks in China
- Regulatory Compliances in Biopharmaceuticals
- Collaborative Opportunities and Partnership for U.S.-China Biopharmaceuticals.
back to top 

The following funding opportunity announcements from the NHLBI or other components of the National Institutes of Health, might be of interest:
NIH Guide Notices:
Program Announcements (PA):
Please note that most links to RFAs, PAs, and Guide Notices will take you to the NIH Web site. RFPs will take you to FedBizOpps. Links to RFPs will not work past their proposal receipt date. Archived versions of RFPs posted on FedBizOpps can be found on the FedBizOpps site using the FedBizOpps search function. Under “Document to Search,” select Archived Documents.
back to top 

The NHLBI Division of Extramural Research Activities (DERA) is pleased to announce the addition of Dr Lawrence Mahan, as the Director of the Office of Translational Alliances and Coordination (OTAC). Dr. Mahan’s professional experience spans academia, government and industry in both basic and applied biomedical research. Additionally it includes global business and strategic alliance development, strategic planning, technology evaluation, entrepreneurship guidance, and consulting on platform technology development in the life sciences.
Most recently Dr. Mahan served as Director of Innovation and Business Development for Children’s National Medical Center and its research institutes, the Children’s Research Institute and the Sheikh Zayed Institute for Pediatric Surgical Innovation, where he managed intellectual property, strategic business alliance development and the advancement of academic entrepreneurship.
back to top 

The D.C. region is home to some 765,000 jobs that require knowledge in science, technology, engineering and math, representing 27 percent of the overall workforce, according to a report released Monday by the Brookings Institution. Only Silicon Valley ranked higher in percentage of STEM labor.
Greater Washington has consistently ranked near or at the top of the nation in STEM job rankings, owing largely to the federal government and the contracting industry surrounding it, which grew rapidly following the Sept. 11 terrorist attacks. The region also boasts a substantial cluster of commercial tech companies - many of them situated along the Dulles corridor and Interstate 270 - as well as a small but growing software startup scene.
back to top 

Almost a quarter of the jobs in the Baltimore-area are science, technology, engineering and math positions, according to a new report by the Brookings Institution.
A total of 281,730 local STEM jobs account for about 23 percent of workforce in the Baltimore-Towson region. Baltimore ranks eighth out of 100 metropolitan areas for its concentration of STEM jobs in the Metropolitan Policy Program study released Monday by the Washington, D.C., think tank.
back to top 

Maryland is No. 1 — or is it No. 7? On third thought, it might be 41st.
Critics and champions of the Free State’s business climate and tax policy have plenty to argue about most days, and all-over-the-place business climate rankings do little to quell the conflict.
The U.S. Chamber of Commerce last month rated Maryland No. 1 for entrepreneurship and innovation, piling on to an Entrepreneur Magazine ranking calling Maryland the best state in which to start a business.
back to top 

Histogenics Corp., a regenerative medicine company that combines cell therapy and tissue engineering technologies to develop highly innovative products primarily for orthopedic tissue repair, today announced the appointment of Peter Greenleaf to Chief Executive Officer.
Bringing over 20 years of experience in the biotechnology industry to Histogenics, Mr. Greenleaf most recently served as President of MedImmune, the worldwide biologics arm of AstraZeneca. During his tenure, he presided over the expansion of MedImmune's extensive growth and pipeline and spearheaded industry-leading business development and venture deals. Mr. Greenleaf also served as the President of MedImmune Ventures, overseeing more than $300 million in investments in early stage portfolio companies. Prior to becoming President, Greenleaf led the development of the company's global marketing and portfolio organizations and managed the broader commercial, corporate development and strategy functions.
back to top 

BioHealth Innovation is working to assist relevant SBIR projects through its Commercial Relevance Program for the August 5th, 2013 NIH SBIR Deadline.
If your company is planning a submission for the August 5 deadline, BioHealth Innovation can assist with your submission. Through the BHI Commercial Relevance Program for SBIR/STTR projects select companies submit their federal funding concepts and receive pre-proposal feedback to help strengthen your application. Further support from BHI’s network of professional consultants and service providers is available to assist in improving your application. If you’re are planning a submission for the August 5 deadline contact Ethan Byler for details on possible support from BHI.
NIH SBIR Phase I Program
- Companies to complete brief application describing the proposed SBIR project apply by June 18th
- All companies notified of SBIR assistance by June 26th
- Guidance, writing strategy, direction, and review sessions to be schedule to enable you through grant submission
- Feedback and redlined comments on written drafts and proposals materials as well as final submission assistance
back to top 

Five to ten years. That’s how long it will be before drug reimbursement in the United States becomes as stringent as in Europe, according to a range of consultants, analysts, and health policy experts with whom I’ve spoken.
This new reimbursement environment – and the expectations leading up to it – is expected to emphasize the value of “profound” innovation, at the expense of less dramatic, incremental innovation.
back to top 
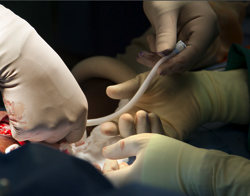
In a first-of-its-kind operation in the United States, a team of doctors at Duke University Hospital helped create a bioengineered blood vessel and transplanted it into the arm of a patient with end-stage kidney disease.
The procedure, the first U.S. clinical trial to test the safety and effectiveness of the bioengineered blood vessel, is a milestone in the field of tissue engineering. The new vein is an off-the-shelf, human cell-based product with no biological properties that would cause organ rejection.
back to top 

NIH has reissued its Small Business Innovation Research (SBIR) Omnibus Grant Solicitation announcement, which states that small businesses that are majority-owned by multiple venture capital operating companies are eligible to apply for (1) these SBIR grants and (2) any other NIH SBIR funding opportunities announced after January 28, 2013. The NIH grant solicitation announcement can be found here.
With this re-issuance, small businesses that are majority-owned by multiple venture capital operating companies (VCOCs), hedge funds and/or private equity firms are now eligibleto apply to the NIH SBIR program and compete for up to 25% of NIH’s SBIR set-aside.
back to top 

Healthcare providers are taking telemedicine to new heights, with the market seeing growth of a whopping 237 percent within a five-year period, according to a new Kalorama report.
Officials say the telemedicine patient monitoring market grew from $4.2 billion in 2007 to more than $10 billion in 2012. According to the report, the market itself is considered small- to moderate in size but makes up for it with its notable number of competitors and "increasing awareness of effectiveness."
back to top 
Investing in biotechnology is a riskier bet these days. The community of life sciences venture capital firms is contracting, despite scientific advances across many fields like g enomics, immunology, and diagnostics. Many promising new enterprises fail to produce marketable drugs, and even successful therapies may struggle to gain markets in an environment of health care cost cutting. enomics, immunology, and diagnostics. Many promising new enterprises fail to produce marketable drugs, and even successful therapies may struggle to gain markets in an environment of health care cost cutting.
That’s exactly why Johnson & Johnson (NYSE: JNJ) chose to expand its programs that nurture very early stage biotechnology and device startups in the Bay area, J&J executives said as they opened the company’s California Innovation Center in Menlo Park, CA, this week.
back to top 
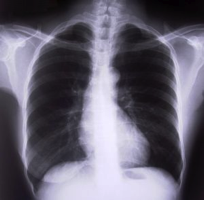
Pathologists and clinical laboratory professionals who regularly analyze images will be interested in the findings of a research study designed to assess how the phenomenon called “inattentional blindness” among radiologists could cause them to possibly miss things hiding in plain sight.
‘Inattentional Blindness’ Occurs Even Among Highly-trained Radiologists
In a recent study, psychological scientists from Harvard’s Brigham and Women’s Hospital found that 83% of radiologists didn’t notice an image of a gorilla embedded in a computed tomography (CT) lung scan.
back to top 

Health Tech Hatch, a site launched last fall as a crowdfunding site specifically for health startups, is joining forces with one of the biggest crowdfunding platforms on the web, Indiegogo.
From the beginning, the company planned to help health startups both crowdfund and beta test their products with patients and physicians. But now, founder and CEO Patricia Salber said Health Tech Hatch plans to focus more closely on the beta testing side, while working on the crowdfunding piece through Indiegogo.
back to top 

San Francisco digital health accelerator Rock Health is kicking off its fifth program next week, and it has some unique startups in the mix.
Bound to be the most talked about is Augmedix, which is developing a healthcare app for Google Glass. Co-founders Ian Shakil and Pelu Tran haven’t said much about exactly what kind of app they’re working on, but it apparently will leverage Glass’s augmented reality and voice activation to help doctors keep their focus on patients. Augmedix has already raised $55,000 from 32 Upstart backers.
back to top 

More than 60 companies showed off their health data applications at Datapalooza IV this week and Krishna Yeshwant, a partner at Google Ventures, was the MC for a demo session on Tuesday afternoon.
As one of the presenters was fighting with his PowerPoint demonstration and the projection system, Yeshwant answered a few questions about opportunities for healthcare startups with the search company’s investment group. He said that the outlook has changed for healthcare IT startups now that Obamacare is here to stay.
back to top 

Through collaboration between Johnson & Johnson Innovation and the California Institute for Quantitative Biosciences (QB3), Janssen will create a 5,000-square-foot lab space within QB3’s 24,000-square-foot incubator space in San Francisco.
The company said that the new Bay Area incubator will use the San Diego model for its innovation space.
back to top 

HHS Secretary Kathleen Sebelius opens Health Datapalooza IV with an emphatic introduction speech asking those in attendance to continue driving more ideas and innovation in data and information exchange to improve the future of the US health system.
back to top 
 ROCKVILLE, MARYLAND, June 4, 2013 – BioHealth Innovation, Inc. (BHI), a regional private-public partnership focusing on commercializing market-relevant biohealth innovations and increasing access to early-stage funding in Central Maryland, is proud to announce the publication of the Central Maryland BioHealth Entrepreneur's Resource and Finance Guide 2013. The Guide was developed by BHI, the Economic Alliance of Greater Baltimore and the Montgomery County Department of Economic Development, in collaboration with the Baltimore Business Journal. ROCKVILLE, MARYLAND, June 4, 2013 – BioHealth Innovation, Inc. (BHI), a regional private-public partnership focusing on commercializing market-relevant biohealth innovations and increasing access to early-stage funding in Central Maryland, is proud to announce the publication of the Central Maryland BioHealth Entrepreneur's Resource and Finance Guide 2013. The Guide was developed by BHI, the Economic Alliance of Greater Baltimore and the Montgomery County Department of Economic Development, in collaboration with the Baltimore Business Journal.
The Guide serves as a compendium of resources to biohealth innovators and entrepreneurs working to start and grow new companies and technologies in the region. This essential entrepreneurial resource includes a compilation of information on financial resources, university facilities and programs, economic development programs, and existing federal laboratory facilities and programs, as well as how to work with these assets. The Guide is one of the many projects BHI is developing to successfully transform the regional environment for biohealth startups through harnessing Central Maryland's biohealth assets and establishing an entrepreneurial ecosystem.
The Guide has been distributed through the Baltimore and Washington business journal subscribers, and is being distributed to all regional partners. For your copy of the guide please download here or contact BioHealth Innovation for details on how to receive a hard copy.
back to top 
Wednesday, June 19, 2013, 06:00pm - 09:00pm
CoFoundersLab & Society of Physician Entrepreneurs proudly presents an educational and networking forum for entrepreneurs in healthcare and life sciences.
To make the most of your time, please browse CoFoundersLab.com and filter by "Meetup Groups - Society of Physician Entrepreneurs" before attending. If you have not created a profile on CoFoundersLab.com, please note that this is required in order for people to discover you before the event and seek you out during the networking portion of the event.
More Information
back to top 
|